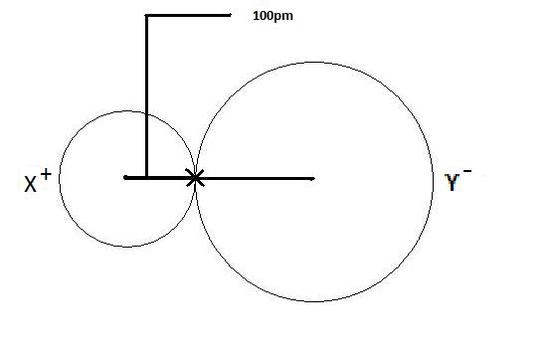
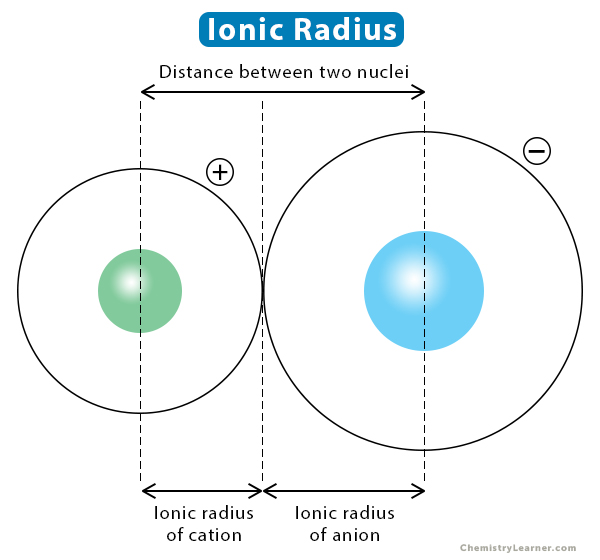
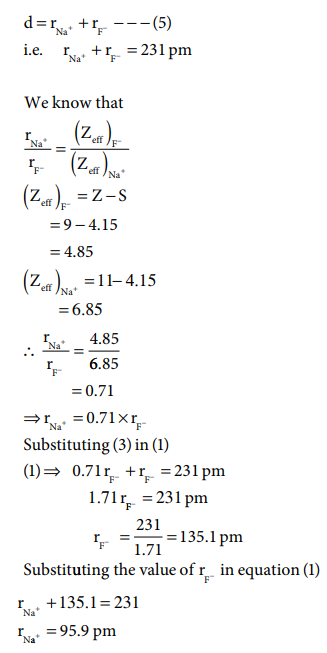Calculation of Ionic radius of Na+&F-/periodic classification of elements/Unit 3/11thNew sy in Tamil - YouTube

Atomic radius of `Li is 1.23 Å` and ionic radius of `Li^(+)` is `0.76 Å`. Calculate the percentage - YouTube

IJMS | Free Full-Text | Theoretical Calculation of Absolute Radii of Atoms and Ions. Part 2. The Ionic Radii
![PDF] Theoretical Calculation of Absolute Radii of Atoms and Ions. Part 2. The Ionic Radii | Semantic Scholar PDF] Theoretical Calculation of Absolute Radii of Atoms and Ions. Part 2. The Ionic Radii | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/75cdc9eb64c74ee1b407825b17bb9dc6ca1c69a7/4-Table1-1.png)
PDF] Theoretical Calculation of Absolute Radii of Atoms and Ions. Part 2. The Ionic Radii | Semantic Scholar
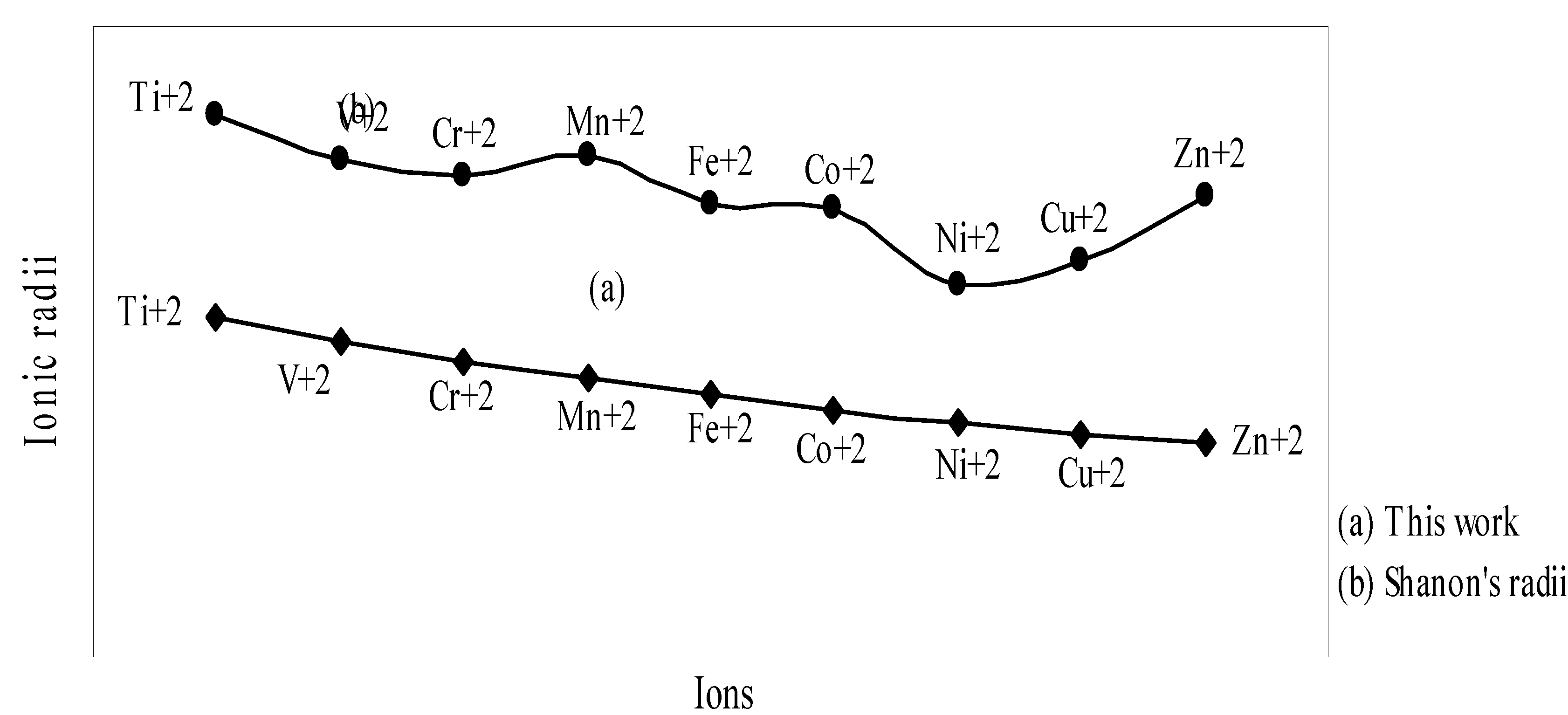
IJMS | Free Full-Text | Theoretical Calculation of Absolute Radii of Atoms and Ions. Part 2. The Ionic Radii

By using Pauling method calculate the ionic radii of Na+ and F- ions in the sodium fluoride crystal - Brainly.in

Atomic radius and ionic radius of `F_(g)` and `F_(g)^-` are `72` and `136` pm prespectivley. - YouTube

Calculate the ionic radius of Cs^+ ion assuming that the cll edge length of CsCl is 0.4123 nm an... - YouTube

Calculate the ionic radius of a Cs +ion assuming the cell edge length for CsCl is 0.4123 nm and that the ionic radius of a Cl ion is 0.181 nm.A. 0.176 nmB.

Ionic Radius Trends, Basic Introduction, Periodic Table, Sizes of Isoelectric Ions, Chemistry - YouTube

![Ionic radii of cation used for calculation of the geometry factors [39]. | Download Table Ionic radii of cation used for calculation of the geometry factors [39]. | Download Table](https://www.researchgate.net/publication/310391155/figure/tbl3/AS:668380013015050@1536365621728/Ionic-radii-of-cation-used-for-calculation-of-the-geometry-factors-39.png)