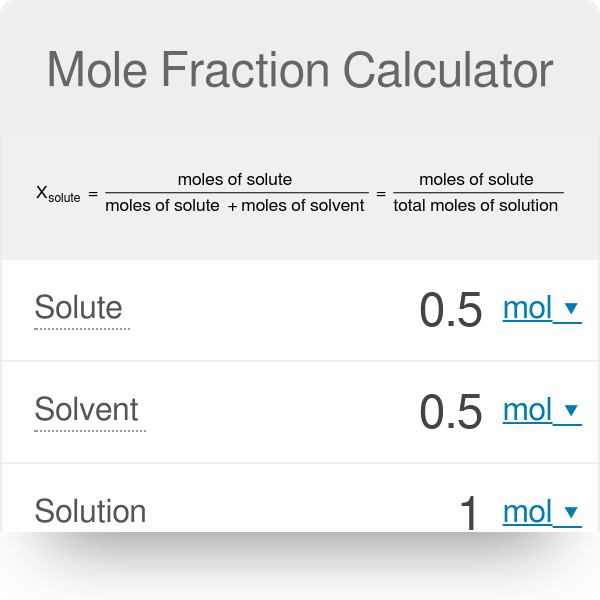
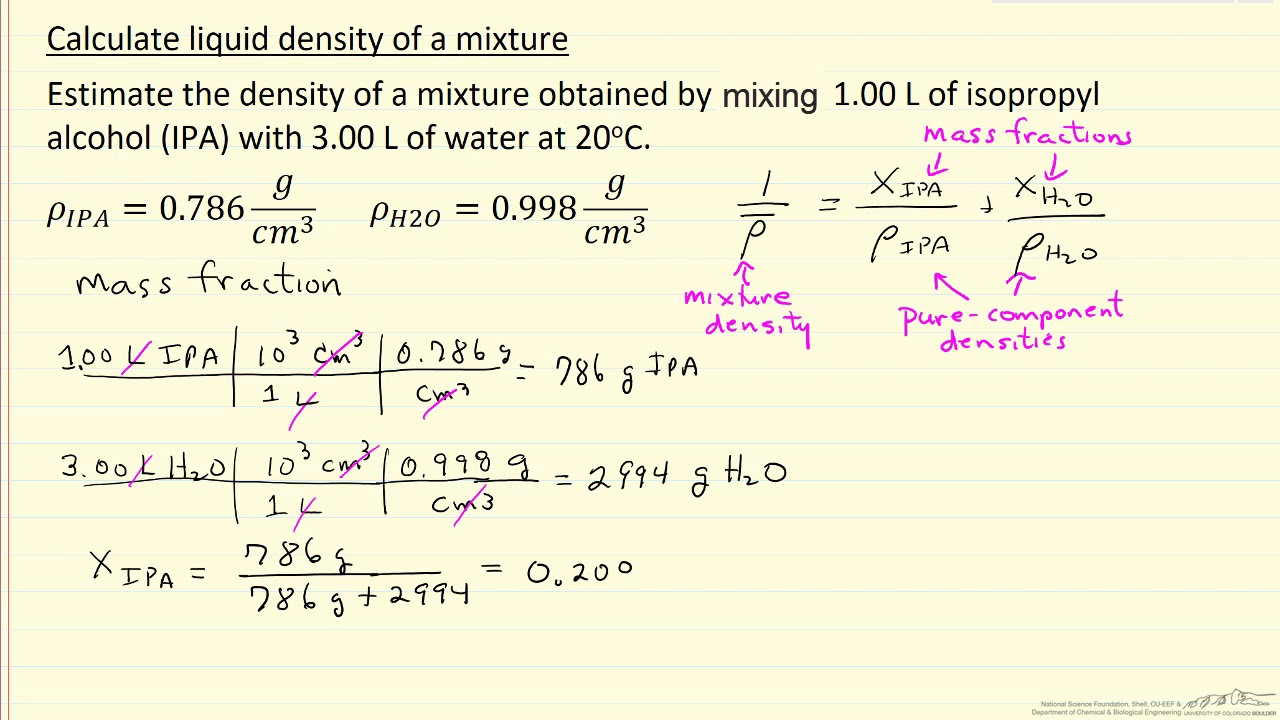Question Video: Determining the Mole Fraction of a Gas Given the Mole Fraction of the Other Gas in the Mixture | Nagwa

Radial profiles of mean mixture fraction, rms fluctuation of mixture... | Download Scientific Diagram

Graph of mole fraction and temperature against mixture fraction from... | Download Scientific Diagram

Flash equilibrium calculation of a mixture - Mixture Bubble point and dew point temperature calculation

LMI calculation: instantaneous value of the local heat release as a... | Download Scientific Diagram

SOLVED: A gas mixture contains 1.25 g N2 and 0.85 g O2 in a 1.55-L container at 18 C. Calculate the mole fraction and partial pressure of each component in the gas mixture.


![Pharma Engineering: [HOW TO] Calculate Density of solvent Mixture Pharma Engineering: [HOW TO] Calculate Density of solvent Mixture](https://2.bp.blogspot.com/-IMT9YlNZa-M/V3ROrtEm7UI/AAAAAAAAAH0/vcCuiz3jQ0MyQfgXYyrDb2TfMC7SARykACLcB/s1600/slide_2.jpg)