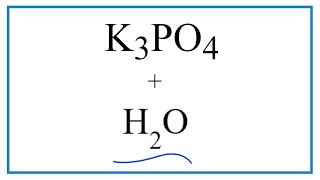
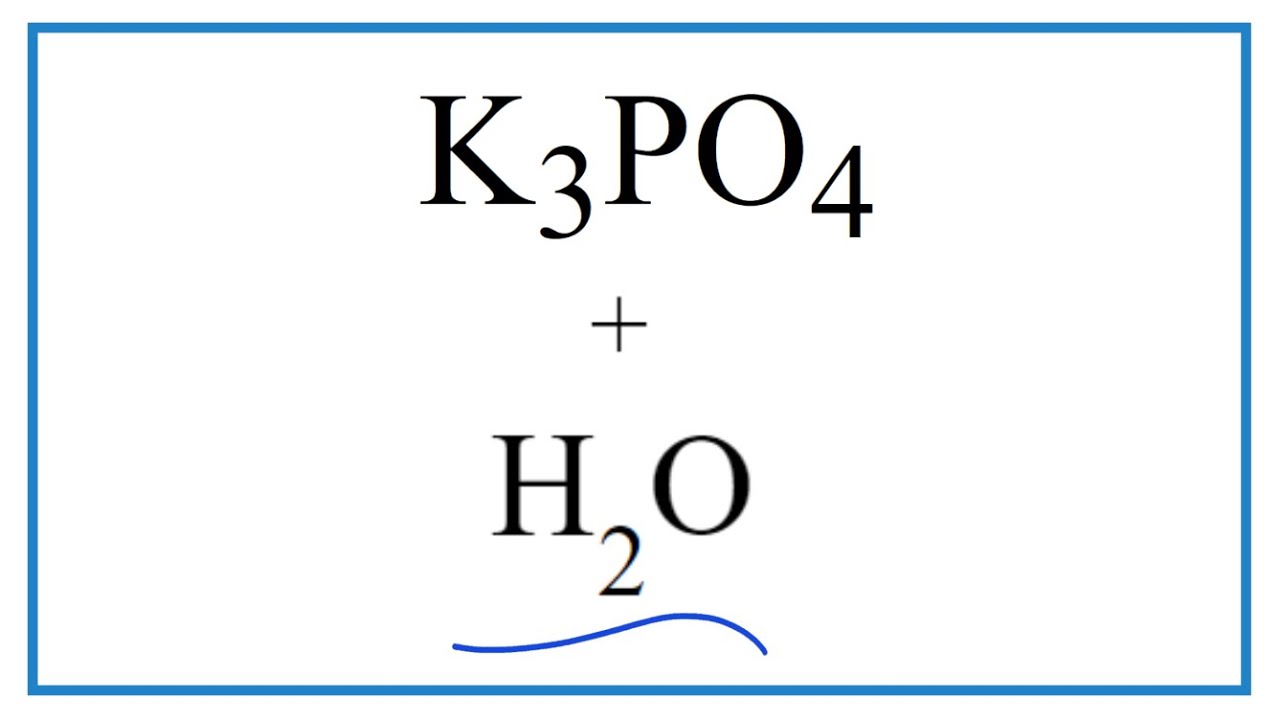
SOLVED: The balanced chemical equation for the reaction of the base potassium hydroxide with phosphoric acid to form potassium phosphate and water is: 3 KOH HPO4 KaPOs 3 HzO How many grams

Unit 11: Equilibrium / Acids and Bases reversible reaction: R P and P R Acid dissociation is a reversible reaction. H 2 SO 4 2 H 1+ + SO 4 2– - ppt download

A NEW METHOD FOR ONE-POT SYNTHESIS OF ARYLOXYPHENOXYPROPIONATE HERBICIDES USING 2,4,6-TRICHLORO-1,3,5-TRIAZINE AND (n-BU)4NI AS A HOMOGENEOUS CATALYST