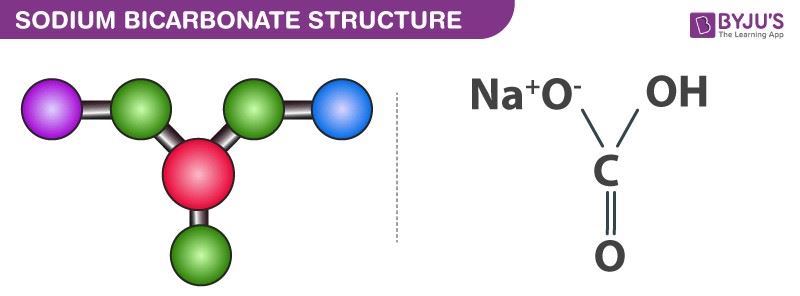
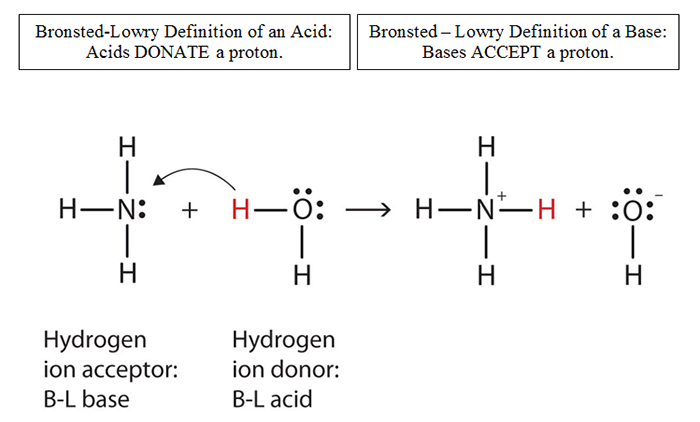
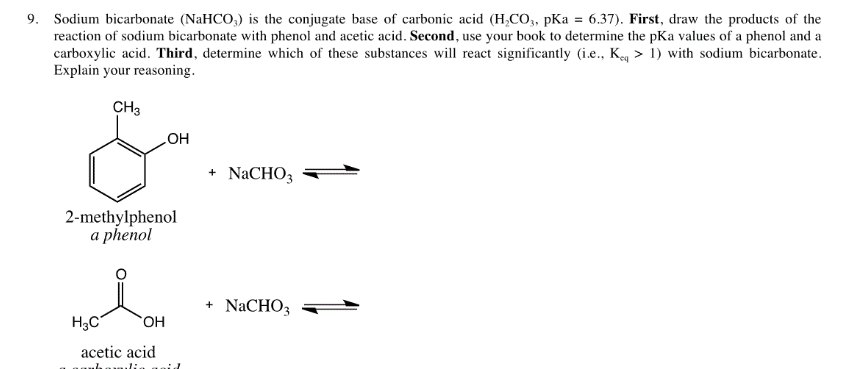
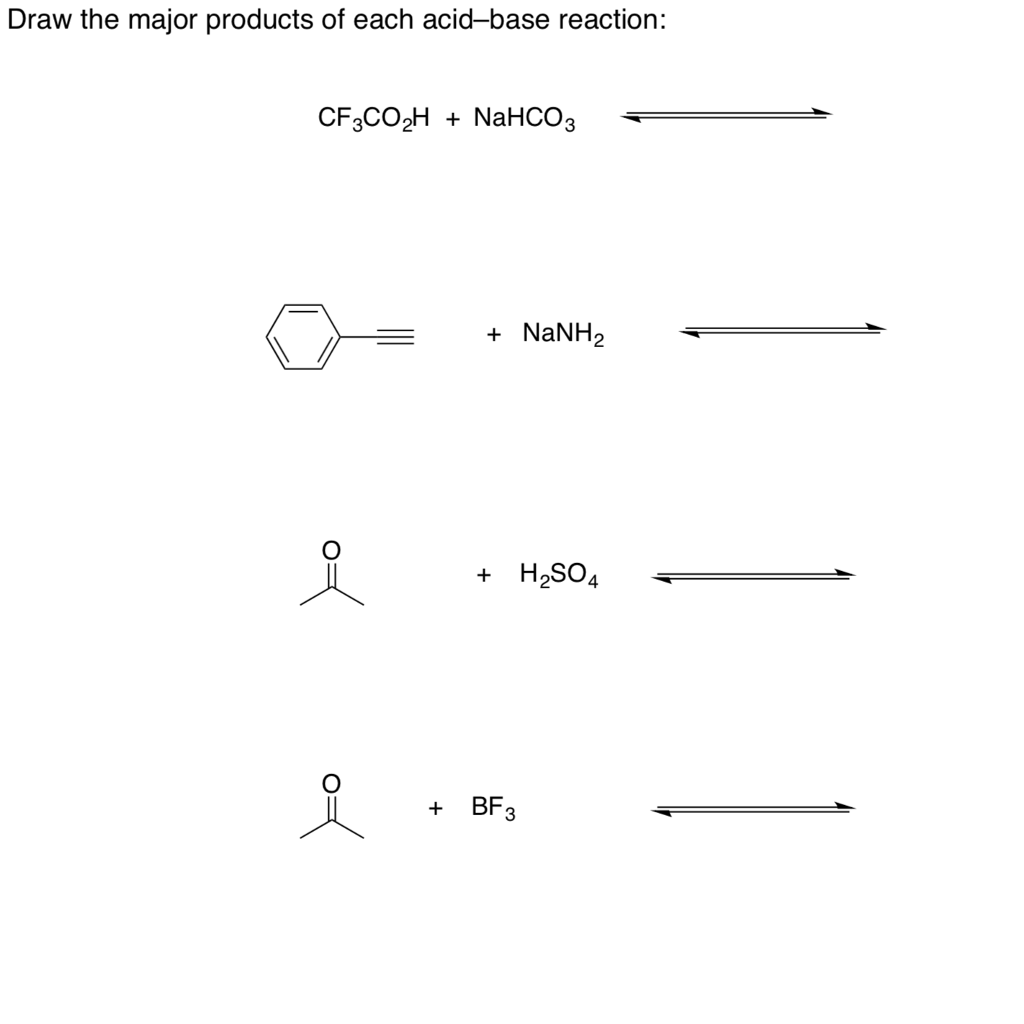
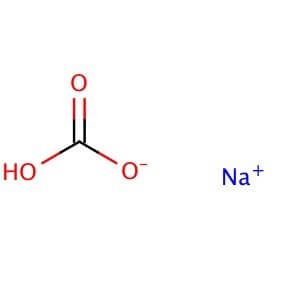
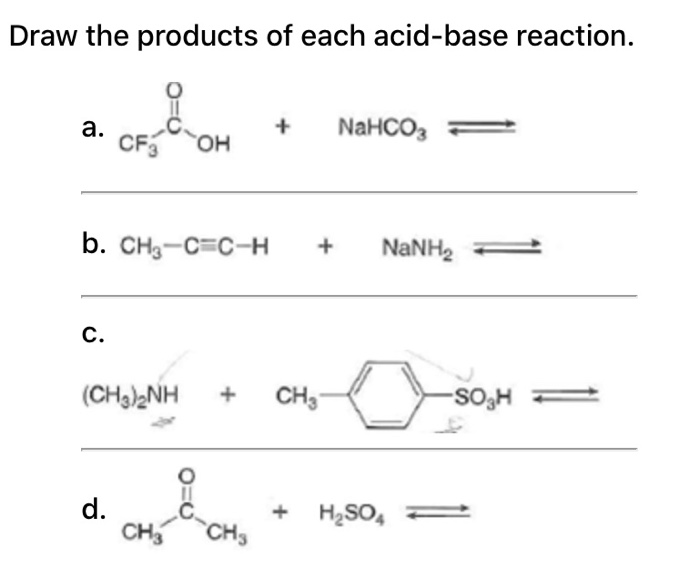
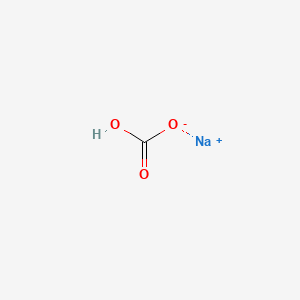
Is NaHCO3 considered a Lewis acid even though Oxygen has a neg charge and lone pairs which easily classifies it as a base?? Please help, I am so confused. Also, since (CH3)3C+
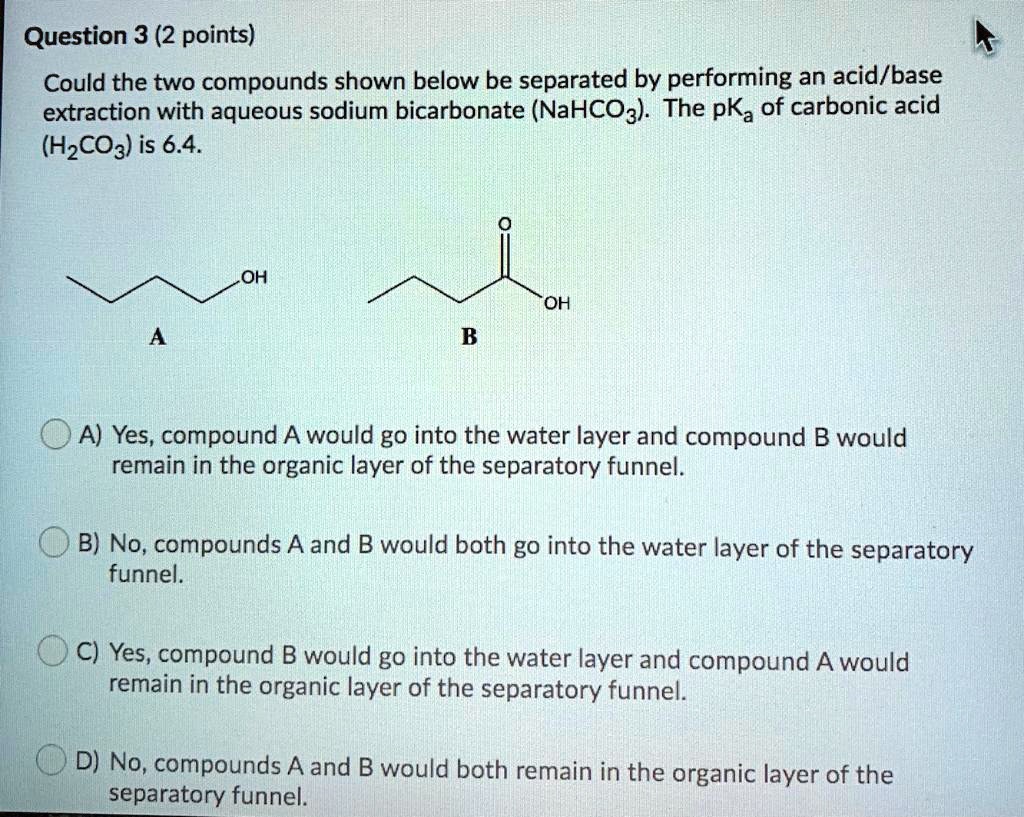
SOLVED: Question 3 (2 points) Could the two compounds shown below be separated by performing an acid/base extraction with aqueous sodium bicarbonate (NaHCO3) The pKa of carbonic acid (HzCO3) is 6.4. OH