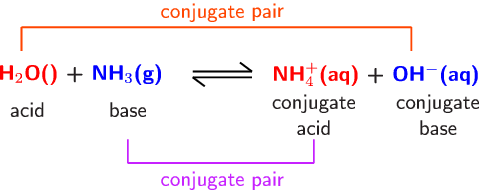
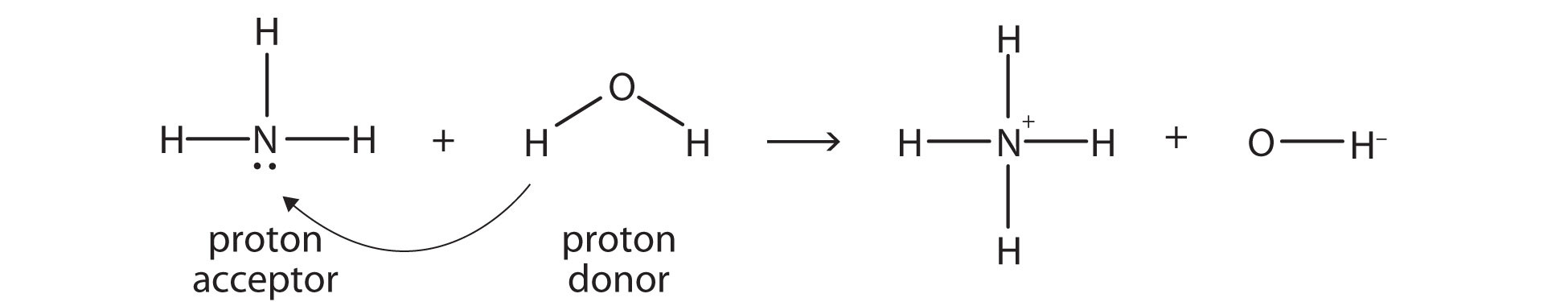
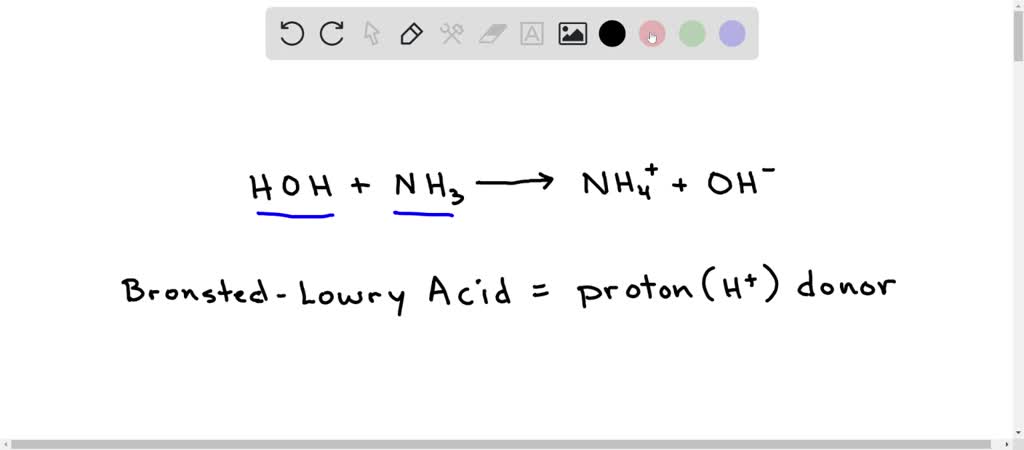
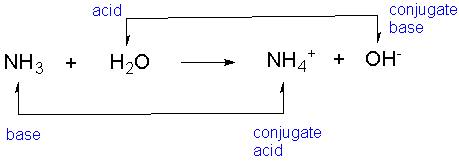
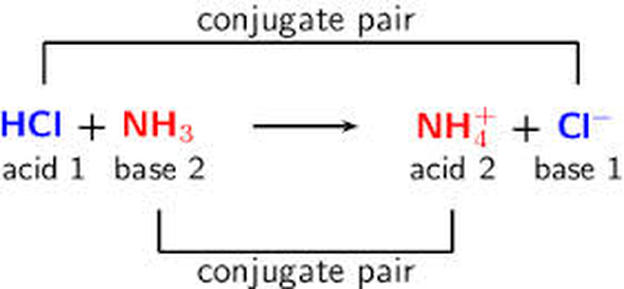
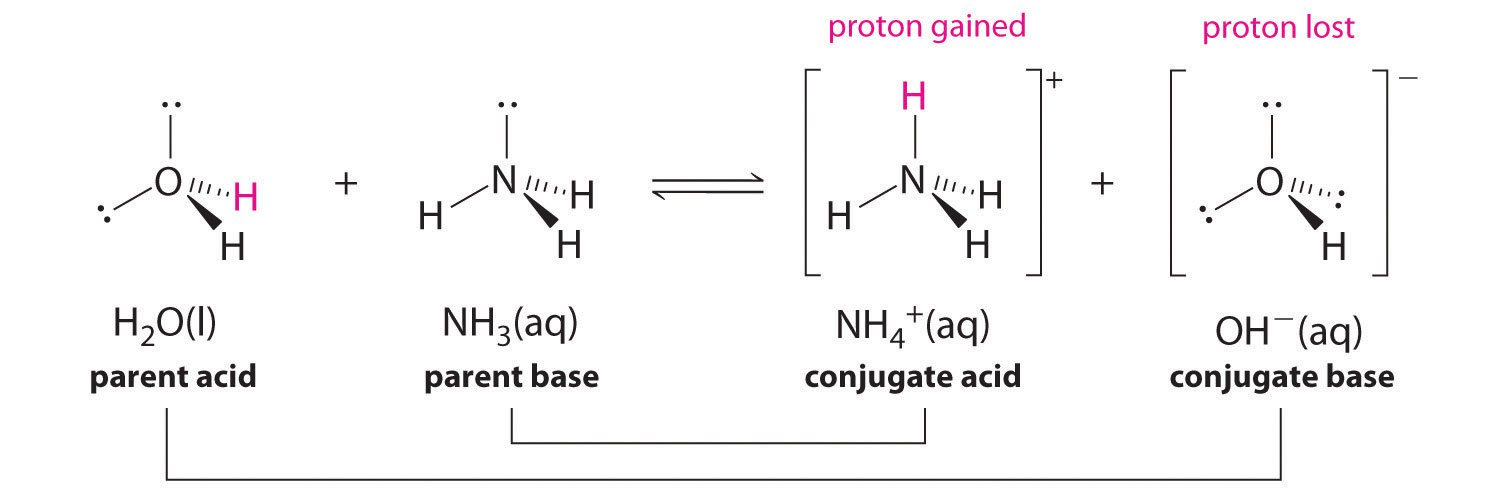
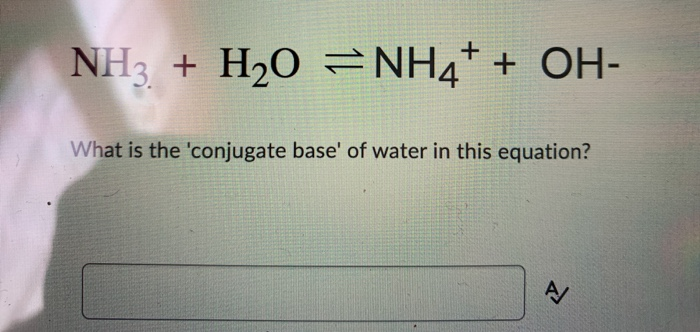An acid-base reaction can occur when ammonia (NH3) and water (H2O) are mixed. Draw the curved arrows depicting the electron flow for the following acid- base reaction. Draw the conjugate acid and

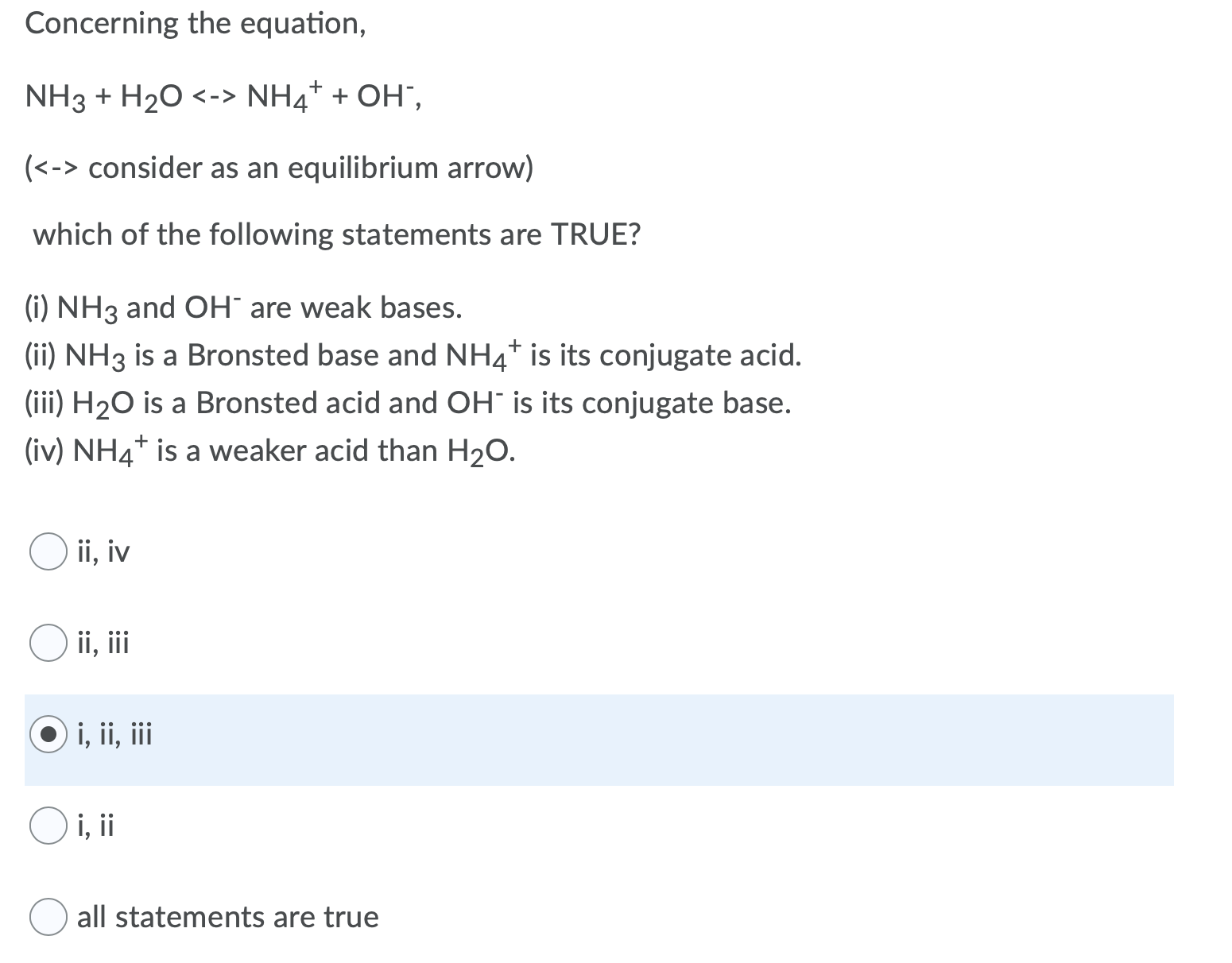
SOLVED: In the following reaction: NH4+ + H2O = NH3 + H3O+ A) H2O is a base and NH3 is its conjugate acid B) NH4+ is an acid and H20 is its